Standardization Allows MRI to Overcome Neuroradiology Challenges
By: Karen Holzberger, President & CEO of SpinTech MRI
From the inception of electroencephalograms to the development of bionic limbs, the world of medical technology advanced significantly in the last century. In the realm of in vivo imaging, magnetic resonance imaging (MRI) is still relatively new, but its versatility and indispensability have made it one of the most important diagnostic tools in modern medicine.
The Role of MRI in Medical Imaging
As the use of MR in a clinical setting evolves, the medical community still relies heavily on topographic imaging means such as CT or PET scans. However, just as there’s a time and place for X-ray or any other type of imaging scan, MRI plays an important role in the medical imaging world.
In Neuroradiology, the highly detailed images produced by MRI are often sought as a necessity when topographic imaging falls short. Alongside the well-known benefit of MRI not exposing the patient to ionizing radiation, the ability to provide contrast to subtle changes in water content or small microhemorrhages carries great diagnostic and research potential.
For a disease such as Multiple Sclerosis where a care provider’s prerogative may require frequent imaging, MRI reduces the ongoing risk of repeated exposure to cellular damage. In the case of a stroke patient, while MRI tends to come after CT due to time constraints, MRI allows us to determine exactly where the blockage is, exactly what brain tissue doctors may be able to salvage, and provides critical features for assessing intervention risks and benefits.
Overcoming MR Criticisms: Standardization
Despite what the name may suggest, “standardization” doesn’t mean that every MR scanner should be the same, or that every patient should receive the same analysis or protocol. Rather, standardization seeks to provide machine-agnostic protocols on which essential and meaningful conclusions can be made.
The purpose of standardization is to increase the direct comparability within MRI, whether from different sites, manufacturers, or databases. The ability to reduce systematic errors which occur due to acquisition dependence will help limit inter-variability in patient scan data, allowing for effective longitudinal comparisons even outside strictly controlled research studies and trials. For clinical operations, this opens up a multitude of avenues for allocating medical imaging resources and offering patients’ logistical choices.
Even with its incredible power, there are several valid criticisms of MRI, but continued advances in supplemental technologies may allow us to eliminate these issues entirely. Below are a few of the common criticisms of MRI technology and how MRI standardization can overcome them.
Consistent Scan/Rescan
The Criticism
Because of the way that MRI is acquired there are many potential areas for variability in the final image. Not only are there different imaging parameters that must be specified to generate the desired image, the ability to manipulate or change these parameters is often limited based on the scanner’s hardware and software. This variance precludes meaningful comparability between different imaging databases, or even between images acquired under the same protocol on MR scanners from different manufacturers.
The Solution
A simple path forward is to perform a scan with the same settings to allow the machine to capture consistent images. In practice, however, MR scanners are not equally calibrated and have unique user traits. Therefore, to ensure that scan/rescan has an optimum outcome when building databases or within multi-site trials, it’s important to assess the hardware participating, then standardize data collection to provide consistent outputs. There are no “bad” scanners, but standardization should consider the potential variability in performance and errors between environments. Methods such as mapping and correcting the magnetic field inhomogeneities will help us to not only see the brains contrast in MRI but to understand the underlying tissue characterization and stability through quantitative imaging.
Post-scan Data Manipulation
The Criticism
Three major components of determining an MR scan’s efficiency are signal-to-noise ratio (SNR), resolution, and acquisition time. Radiologists and physicists have experimented over the years on how to achieve the highest-detail output in the shortest amount of time. While many methods may speed up the image acquisition process, they can cause data loss along the way. To make up for this, radiologists will apply various filters to make certain aspects of the image more clear. However, this post-scan manipulation can lead to issues like blurring other potentially important details in the scan.
The Solution
This has proved to be an exciting and dynamic time for MRI, and as with the need for understanding of how different acquisition methods have some degree of error, the same is true for the rapidly evolving field of post-data manipulation in MRI. By implementing standard acquisition and post-processing procedures, radiologists can easily acquire and enhance images for a wide variety of use cases while ensuring that all captured images meet the same criteria for image detail. These protocols have begun to rely on AI assistance to automatically detect relevant biomarkers to aid in diagnosis.
Time Constraints
The Criticism
As previously mentioned, long scan time is arguably the biggest downside of MRI, especially in the acute setting. A radiologist could theoretically spend three hours with a patient acquiring one incredibly detailed scan with a protocol more aptly suited for a cadaver, but we run into a circumstance of diminishing returns. Those three hours would be, arguably, better-spent acquiring a variety of shorter, data rich scans that provide a greater breadth of information for diagnosing the patient. Indeed, the long-scan times in MRI also mean that it is at times plagued by motion artifacts which are detrimental to detailed and quantitative images. For MR centers, high-throughput often means acquisition time for a protocol must be 15 minutes or less, and motion affected images may not be repeated.
The Solution
Motion-tracking and correction during acquisition, post-processing registration, and AI combined with acceleration techniques all have the potential to mitigate patient motion in MRI. In addition, rapid multi-parametric imaging has the potential to provide inherently co-registered images which could be used in combination with the above techniques. With so many emerging choices in MRI, standardization seems like a distant goal, however taking steps towards standardization by utilizing new technologies will help create efficiency and benefits not only from chipping away protocol time, but by improving the available information to enhance the patient-provider decision making process.
High Costs
The Criticism
Like most pieces of diagnostic equipment, new MRI machines are expensive. Factoring in ongoing maintenance costs, additional hardware, and the cost of building rooms with adequate electromagnetic shielding, facilities with MR scanners need to be constantly imaging patients to make up for those costs.
The Solution
Decreasing the necessary time per scan means seeing more patients every day, helping medical facilities offset some of the costs of MRI. Replacing and adding scanners is also a high-cost investment, therefore taking advantage of emerging standardized technologies means compatibility with both new and aged systems. This means your facility can go longer without substantial capital investment in new equipment while improving the quality of your MRI scans.
The Strongest Future Medical Imaging Tool
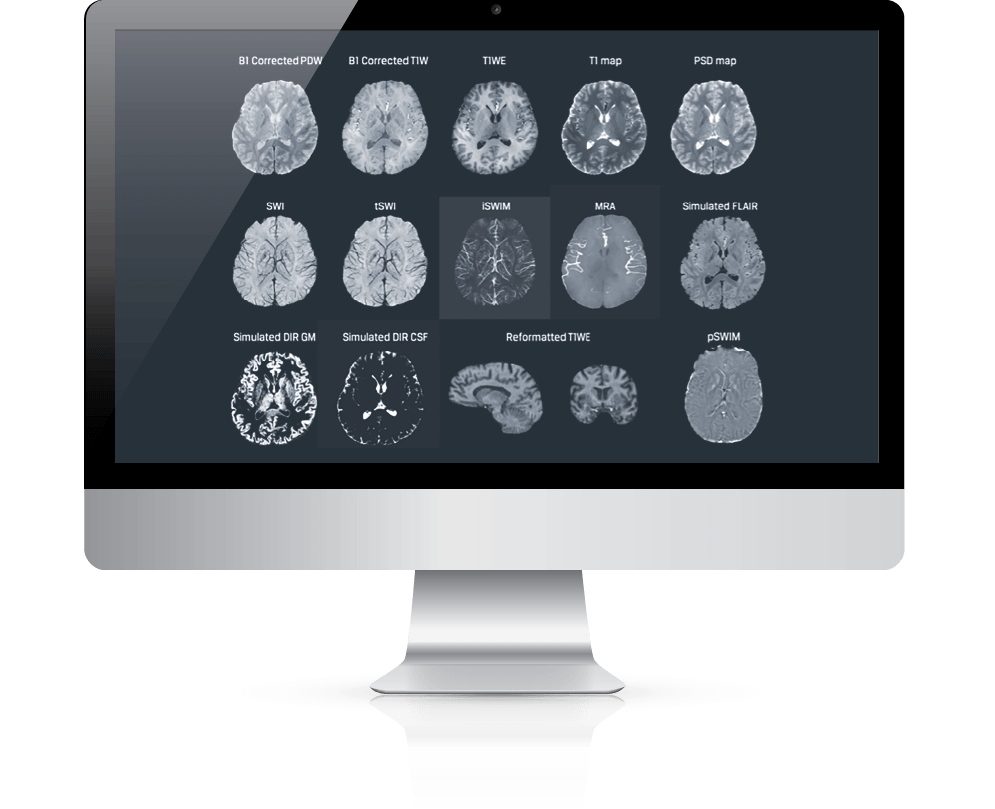
The ability to standardize MRI isn’t some nebulous, futuristic wish: the technology already exists. Supplementary technologies like STAGE allow users to standardize the approach to the process, simplifying the work of radiologists and arming neurologists with straightforward, easy-to-interpret images for improved, streamlined patient care. With high-resolution, quantifiable images, doctors can see and measure in real-time the effects of disease progression and therapy on the body to a significantly higher level of detail than topographic scans.
As we standardize quantitative MRI in our research consortiums, collaborative databases and within the clinical setting, it could potentially make MRI the most powerful imaging tool for neurologists in the not-so-distant future.

