Susceptibility-weighted Imaging: Technical Essentials and Clinical Neurologic Applications
By: Karen Holzberger, President & CEO of SpinTech MRI
Author(s): Sven Haller, MD, MSc, • E. Mark Haacke, PhD • Majda M. Thurnher, MD • Frederik Barkhof, MD, PhD
Journal: Radiology
Published: 2021
Read Full Paper: https://pubs.rsna.org/doi/abs/10.1148/radiol.2021203071?journalCode=radiology
Abstract
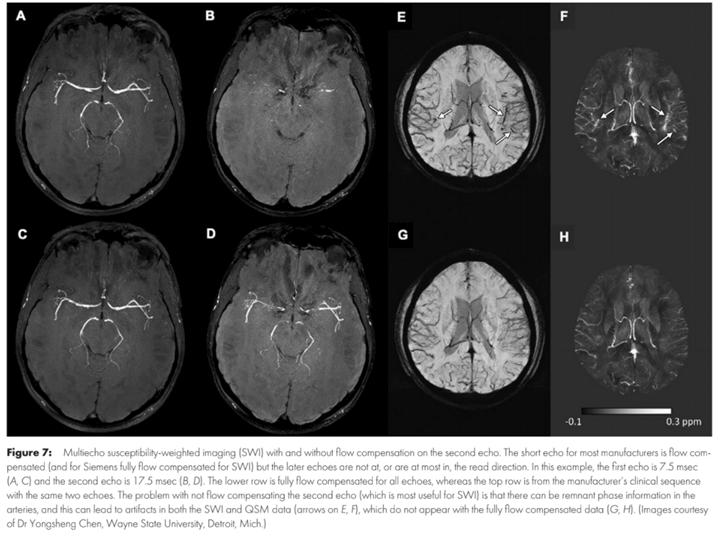
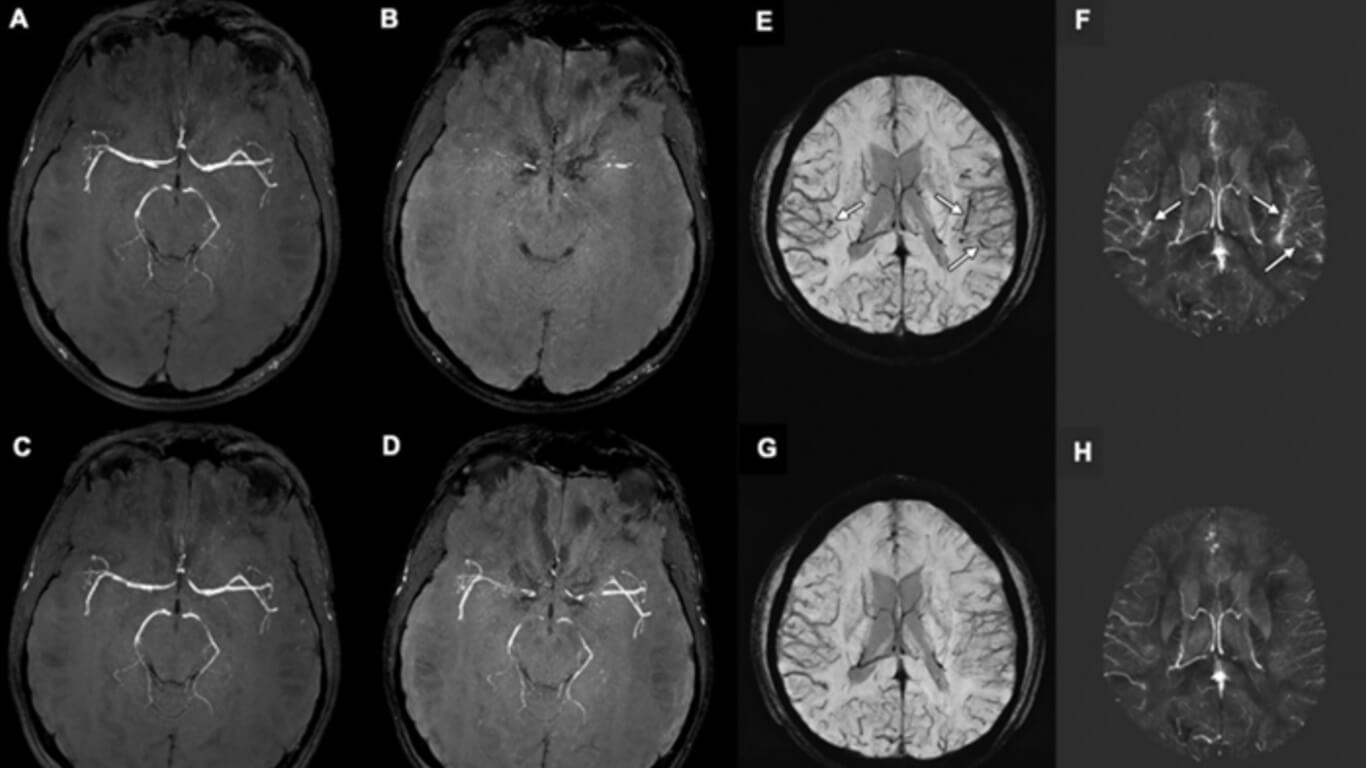
Susceptibility-weighted imaging is an increasingly important adjunct in diagnosing a variety of neurologic diseases and provides a powerful tool to depict and help characterize microbleeds, veins, and other sources of susceptibility. Susceptibility-weighted imaging (SWI) evolved from simple two-dimensional T2*-weighted sequences to three-dimensional sequences with improved spatial resolution and enhanced susceptibility contrast. SWI is an MRI sequence sensitive to compounds that distort the local magnetic field (eg, calcium and iron), in which the phase information can differentiate. But the term SWI is colloquially used to denote high-spatial-resolution susceptibility-enhanced sequences across different MRI vendors and sequences even when phase information is not used.
The imaging appearance of SWI and related sequences strongly depends on the acquisition technique. Initially, SWI and related sequences were mostly used to improve the depiction of findings already known from standard two-dimensional T2*-weighted neuroimaging: more microbleeds in patients who are aging or with dementia or mild brain trauma; increased conspicuity of superficial siderosis in Alzheimer disease and amyloid angiopathy; and iron deposition in neurodegenerative diseases or abnormal vascular structures, such as capillary telangiectasia. But SWI also helps to identify findings not visible on standard T2*-weighted images: the nigrosome 1 in Parkinson disease and dementia with Lewy bodies, the central vein and peripheral rim signs in multiple sclerosis, the peripheral rim sign in abscesses, arterial signal loss related to thrombus, asymmetrically prominent cortical veins in stroke, and intratumoral susceptibility signals in brain neoplasms.

